Manufacturer In China
High chemical activity and excellent catalytic and photocatalytic activity
Yosoar focuses on the production and research of zinc oxide
Kunshan yosoar has been committed to the promotion as well as the overall applications of zinc oxide for a long time.
After years of hard work, it has produced various types of zinc oxide with different morphology, specific surface area, whiteness and other requirements under the condition of meeting market requirements.
Yosoar is the best choice for zinc oxide manufacturer.
It can be used for the production of food-grade materials
It has low content of heavy metals, effective antibacterial
Animal feed grade Zinc oxide is an important material for feed
It can be diluted 200-300 times for antibacterial application
Used in Personal care products
Because zinc oxide has antibacterial and deodorant effects, it can be used in medical ointments to treat skin irritation, and it can also be used in baby powder and creams.
It can also be used in sunscreens, which can reduce sunburn and skin aging, mainly because of the reflective properties of ultraviolet radiation. It can be used as a filler and colorant in many over-the-counter drug products. Paul sunscreen.
In other cosmetics, there are personal care products, including mineral cosmetics and nail products.

Used in antibacterial
Nano-zinc oxide has some good antibacterial properties. Compared with other metal oxides, it has potential biocompatibility, so it can be widely used and can be used in the field of biomedicine.
Nano-zinc oxide has a cytotoxic effect on cells at the same concentration, but is non-toxic to human cells. Due to its ion shedding ability, ZnO NPs show higher toxicity to bacterial cells than other metal oxide NPs (such as TiO 2) effect. Therefore, nano-zinc oxide as a good candidate for nano-dentistry is also rapidly gaining popularity.
Yosoar: zinc oxide nanoparticles Supplier in China
- Certification
- Manufacturing
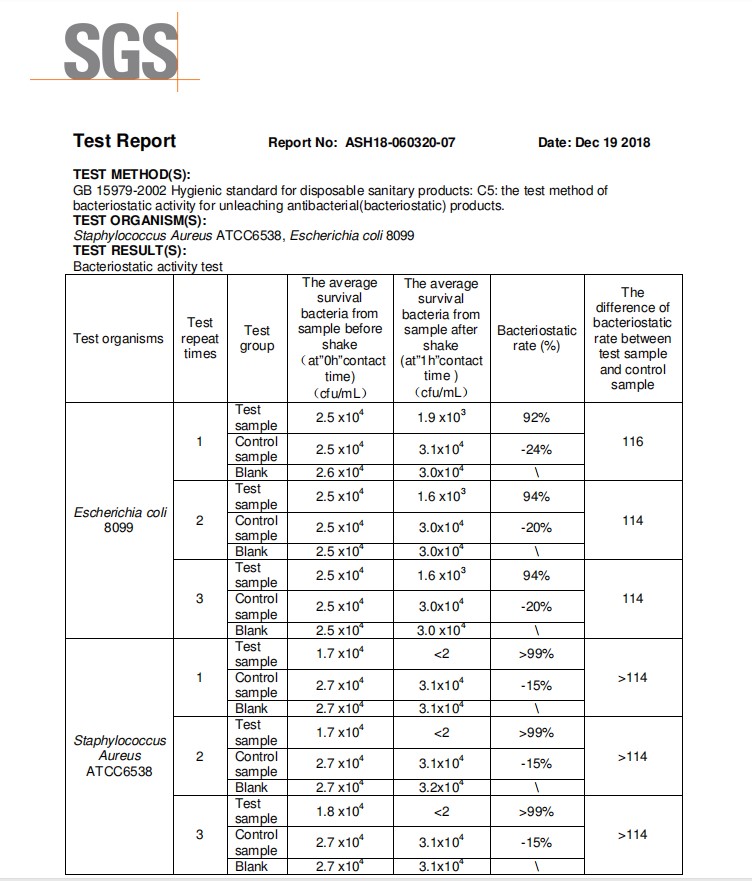
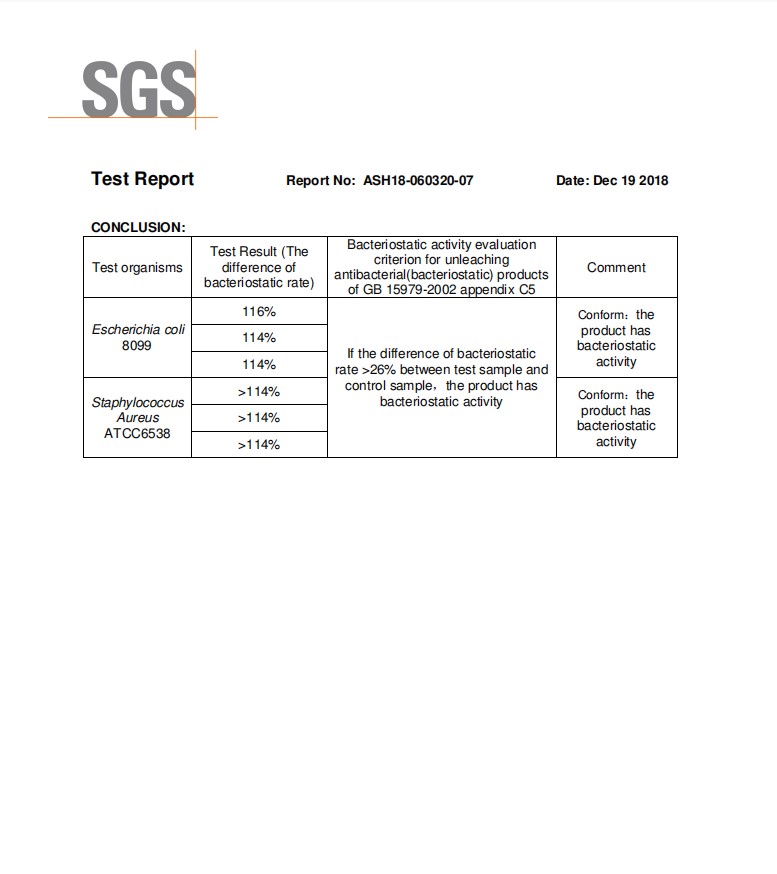
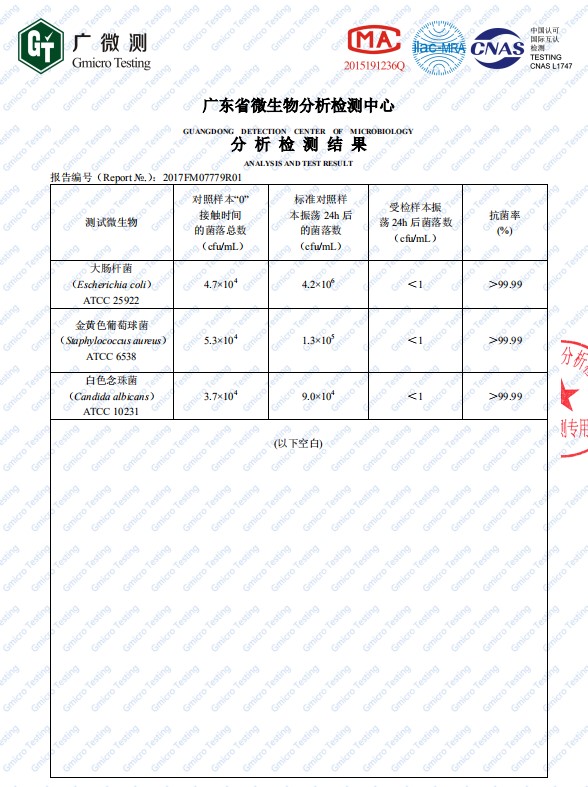
Zinc oxide has a low price, good stability, and abundant sources. It is said about zinc oxide that has become a hotspots in the general research of inorganic antibacterial agents. Studies have shown that nano- ZnO has a strong inhibitory effect on some bacteria such as the Escherichia coli and Staphylococcus aureus, and can inhibit the growth of prokaryotic cells, eukaryotic cells and Escherichia coli in vitro. Under light conditions, the antibacterial effect of zinc oxide powder is more obvious. At present, the research on the principle of antibacterial mechanism of zinc oxide materials is not clear. There are currently three antibacterial principle hypotheses, namely photocatalytic antibacterial, zinc ion elution antibacterial and active oxygen antibacterial mechanism. Extensive research and studies have come up to the conclusion that the overall antibacterial properties of zinc oxide increase with the overall increase of the specific surface area, and decrease with the general increase of the size of the particles. The antibacterial performance of zinc oxide when compared to the ordinary present zinc oxide is generally much better.
With the increasing number of industrialized pollutants, the environmental problems faced by mankind have become more and more serious. In recent years, it has been discovered that some semiconductor materials such as TiO2 and ZnO can be used to degrade pollutants under light conditions. Because of its excellent photocatalytic activity, zno nanoparticles can effectively convert harmful substances into inorganic substances such as H2O and CO2, and some difficult-to-degrade substances can also be effectively removed. Moreover, the price is low, the stability is good, and the toxicity is low, and it has huge application potential in pollution control. Shang Xiangshu used organic dyes as photocatalytic objects and used ellipsoidal nano-crystal particles for photodegradation experiments. The study found that the photodegradation effect is obvious and the sample size and morphology have a greater impact on the photocatalytic effect.

Studies have shown that zinc oxide, as a vulcanization accelerator, has an important effect on the crosslinking speed of chemical bonds, the type and number of crosslinks during the vulcanization process, and then plays a role in accelerating vulcanization. Due to the general features and characteristics of small particle size and large specific surface area, zinc oxide nanoparticles’s unique small size effect and surface effect result in its dosage is only 30%-50% of ordinary zinc oxide. The excellent UV absorption properties of zinc oxide can also significantly improve the light-aging resistance of rubber products. As an additive of rubber products, zinc oxide nanoparticles can improve the flexibility, wear-resistance and high cleanness of rubber products. Can greatly save costs and extend product life.
Ultraviolet rays have a strong penetrating ability and have a cumulative effect. Long-term sun exposure can cause photo-aging phenomena such as rough skin and increased wrinkles. The nano zinc oxide in the zinc oxide sunscreen is a broad-spectrum ultraviolet shielding agent. Due to its high chemical stability and good safety, it has gradually been used in sunscreen cosmetics. Also, zinc oxide powder has excellent antibacterial effects. It can produce astringent and anti-inflammatory effects when used in cosmetics and has the effect of absorbing skin secretion oil.
Mixing the zinc oxide nanoparticles film formation refers to a film matrix polymer molecule in a mixed film formation under the given conditions. For chemical polymer film matrix materials, the base material is generally mixed with zinc oxide in a certain proportion in a molten state or a solution state to form a film. For natural bio-macromolecule materials with good film-forming properties, generally, the zinc salt solution is mixed with the film substrate solution in a certain proportion, after the solvent evaporates to form a film, it is immersed in an alkaline solution under certain conditions to be converted into nano Composite film of nano zinc oxide.

Along with the general size of the particles of the zinc oxide nanoparticles have significant antibacterial properties shadow ring, the effect of time and concentration of zinc oxide particles that are of nano size and bacteria between its antibacterial properties there is some influence. He Linhai et al. conducted a study and research on the effect of different mass fractions of zinc oxide nanoparticles on Candida albicans. The results showed that, with the rising mass fraction of zinc oxide nanoparticles, antibacterial rate increasing trend becomes gradually slow, description of its antibacterial zinc oxide content is not linear. Also, Ma Zhanxian et al. studied the relationship between different action times and their antibacterial rate and found that with the extension of the action time, the antibacterial efficiency of zinc oxide nanoparticles increased significantly.
It can reduce the water penetration and slow down the growth rate of the inoculated Lactobacillus embryo; the solubility, water content, water vapor transmission rate and also some of the other parameters of the membrane are low, and the composite membrane has zero transmission of ultraviolet light and can absorb more than 80 % Of near-infrared light.
The coating method means that the zinc oxide is uniformly dispersed in a solvent and then the suspension is coated on the overall surface of the material that is being packed. After the solvent evaporates, the packaging material with zinc oxide nanoparticles fixed on the surface is obtained. Radiation and ultrasound are also commonly used in this process. And other auxiliary means.
The preparation methods are diverse and the particle size and morphology can be controlled stably; inorganic materials have better stability than organic materials; they have a wider antibacterial spectrum; the production cost is lower than silver and other precious metals, and the cost is well controlled; Explained by the human body as zinc ions, it will not accumulate in the body like silver or titanium dioxide and cause harm; it is less cytotoxic and has wide range of applications such as in lotions, lotions, ointments, etc. in addition to food packaging; while nanosilver is used for disinfection When the agent comes into contact with the skin, it is easy to cause argyrism, silver poisoning and so on.
Zinc oxide is indeed one of the wide bandgap semiconductor oxide and has a bandgap similar to that of TiO2 and has a strong photocatalytic effect. The light conditions can affect its antibacterial performance. Studies have shown that under conditions of no light zinc oxide of Bacillus subtilis inhibition rate reached 98%, then the light conditions better; and the zno nanoparticles under the illumination of Escherichia coli antibacterial effect is better than the absence of light, and The antibacterial effect of this zinc oxide is much better than that of TiO2 and SiO2.

Detecting bacteriostatic ring method ordinary zinc oxide, zinc oxide nanoparticles, zinc oxide whisker four three Zn – oxide antimicrobial activity, the results showed that: four zinc oxide whisker antibacterial effect on Escherichia coli best, followed by For ordinary zinc oxide, ordinary zinc oxide has the lowest antibacterial activity, and the unique structure and morphology of tetrapod zinc oxide whiskers are one of the reasons for its excellent antibacterial effect; light conditions also have a certain influence on the overall properties related to antibacterial characteristics of zinc oxide, three kinds of antibacterial effect of the zinc oxide in the simulated sunlight better than the absence of light; also, the same kind of zinc oxide has antibacterial effect differences of different species, three kinds of zinc oxide antibacterial effect on Escherichia coli are better than gold Staphylococcus aureus, these indicate that the light conditions affect the antibacterial type and performance of zinc oxide.
Zinc ion dissolution antibacterial Zinc ion (Zn2+) dissolution and sterilization mechanism believes that zinc oxide leads to the dissolution of cell contents by destroying bacterial cell membranes, hindering the synthesis of beneficial bacterial metabolic enzymes, destroying genetic factors, and causing bacteria to lose their biological activity Wait to complete the sterilization process.
Zinc oxide can slowly release Zn2+ in an aqueous medium, while the surface of the bacteria is negatively charged because of the occurrence or the presence of free -COOH. Zn2+ is adsorbed on the overall surface of the bacteria by Coulomb gravity, which damages the bacterial cell wall; also, zinc oxide can interfere with the synthesis of peptidoglycan, hinder the formation of the cell wall, and inhibit cell reproduction and growth. After that, Zn2+ further penetrates the cell wall, replaces the position of cations on the cell membrane surface, binds to proteins or other anionic groups, and disrupts membrane metabolism. And structure; the combination of zinc oxide with -NH2 at the end of chitosan can also increase the general permeability of the bacterial cell membrane; under the suitable pH conditions for bacterial growth, the zinc oxide nanoparticles is positively charged, and there is static between the zinc oxide and the bacteria The effect causes the two to be tightly combined, and as a result it causes damage to the bacterial cell membrane; these will cause the cell membrane to lose its original biological function, which will lead to the outflow of bacterial cytoplasm, and ultimately lead to cell death in order to achieve the purpose of antibacterial characteristics.
Zinc ions and in vivo bacterial proteins, nucleic acids present in the mercapto group (-SH), amino (-NH : ) sulfur-containing, nitrogen-reactive functional groups of the protein denaturation, loss of cell synthase activity. Enzymes are the catalysts of all organisms, which control microbial biochemical reactions. Once enzymes are inactivated, they will cause loss of catalytic performance, making the biochemical reactions catalyzed cannot proceed normally, and affecting other related biochemical reactions, leading to microbial energy metabolism and Material metabolism is blocked, inhibiting the normal reproduction, growth, development and other processes of bacteria.
Reacts with DNA, destroys the normal activities of the functional system in the cell, and hinders the normal progress of metabolism. But when the bacteria are killed, Zn2+ will be released from the bacteria, and then contact with other bacteria, repeat the above anti-bacterial activities, once again showing excellent antibacterial performance, so the antibacterial performance of zinc oxide is very good Continuity. Zn2+ dissolution sterilization does not require light, and the binding force of zinc with sulfur and nitrogen is particularly strong, so zinc oxide shows high antibacterial activity.
Studies have shown that 5 mmol/L concentration of the presence of zinc oxide nanoparticles have a very good inhibitory effect, the inhibitory rate can reach 90%, and 5 mmol/L of ZnCI2 does almost not exhibit antibacterial activity, and no To obtain a more ideal antibacterial effect, it all shows that the influence of Zn2+ dissolution concentration on the general properties related to antibacterial characteristics of zinc oxide is not obvious. The lower concentration of Zn2+ not only stops and inhibits the growth of bacteria but sometimes also promoted the growth of bacteria. Therefore, the antibacterial mechanism of zinc ion dissolution may not be the main antibacterial mechanism of zinc oxide, but an auxiliary effect.
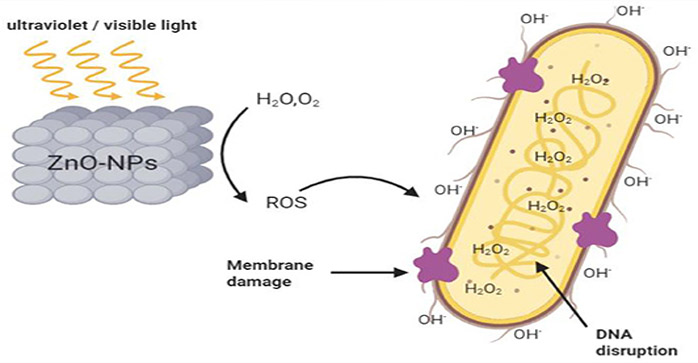
The antibacterial activity of active oxygen produced by zinc oxide is determined by electron spin resonance. It is found that zinc oxide in the zinc oxide solution can stimulate the production of active hydroxyl groups.The active hydroxyl group can further generate other active oxygen groups such as H2O2 through a multi-step reaction. The most significant feature of reactive oxygen species is that they can cause oxidative stress, damage DNA, cell membranes, and proteins, and even cause cell death. Among them, the active hydroxyl group is the strongest active oxygen group known so far, which can react with almost every molecule in living cells. The most important reaction is that two active hydroxyl groups react with each other to generate H2O2.

The antibacterial ability of zinc oxide has a direct relation as these properties increase with the increase of the general specific surface area and concentration of zinc oxide and decreases with the increase of the particle size of zinc oxide, and the degree of influence of the concentration on the antibacterial ability of zinc oxide exceeds the particle size.
The overall effect of light on the antibacterial activity of zinc oxide active oxygen?
Studies have shown that the production of active oxygen is related to light. If we see it with respect to the semiconductor properties and characteristics of zinc oxide, the light will cause the generation of free electrons and electron holes. The water molecules produced -OH and H+ under the action of zinc oxide, and then under the action of dissolved O2, the surface of zinc oxide nanoparticles undergoes a multi-step reaction to produce various active oxygen groups such as H2O2. zinc oxide and an antimicrobial active oxygen photocatalysis antibacterial phase network system.
What is the evaluation of the safety of zinc oxide nanoparticles as food packaging?
Compared with micron-level materials or macroscopic materials, nano-materials are easier to pass through the network structure of the packaging material matrix into the food. Due to the very small size of the nanoparticle, after it enters the body, it can move to the surrounding tissues or even farther, and even break through the blood-brain barrier, forming potential toxicity. Therefore, there has been controversy about the toxicity of nano-sized zinc oxide particles. Relevant studies have shown that zinc oxide nanoparticles particles have cytotoxicity to human bronchial epithelial cells, human lung epithelial cells and human kidney cells. However, some studies show that zinc oxide particles will not enter normal cells or harm the skin of humans or animals. . Nanoparticles in packaging materials enter the human body mainly through the digestive tract. Therefore, for zinc oxide nanoparticles particles in antibacterial food packaging, we must first consider whether they can migrate from the packaging materials into the food. Existence, we need to explore its toxic and side effects on the human body by conducting more experiments that can either be in vivo or either in vitro.
The problems faced in the process of preparing zinc oxide nanoparticles by solid-phase method?
If we consider the overall preparation process of the solid phase method, since no liquid solvent is added, the zinc oxide are mainly prepared by physical methods such as grinding and calcining, so that the pollution to the surrounding environment can be significantly reduced. However, there are disadvantages such as larger sample size, uneven size distribution, and higher energy consumption.
What are the advantages and disadvantages of preparing zinc oxide by liquid phase method?
In the preparation process of the liquid phase method, the solution chemistry method and the direct precipitation method are relatively simple to operate, and the prepared nanoparticles have a small size and uniform distribution, and can be mass-produced, and have a certain potential for expanding industrialization. However, there are still disadvantages such as pollution to the environment during the waste liquid treatment process, easy cause particle agglomeration and easy mixing of impurities during the post-treatment process of the sediment.

After understanding some of the antibacterial mechanisms of zinc oxide, can we use it in large quantities?
The antibacterial effect of zinc oxide is the result of several different mechanisms, and further research and confirmation are needed to explore the mechanism and which mechanism occupies the main position. Apart from this, another fact is that it is precise because the antibacterial mechanism of zinc oxide is not very clear, and it remains in the environment after a large amount of use, so there is a certain threat to the human living environment. We should use it with caution while maintaining experimental research.
What is the relationship between the antibacterial properties of the slurry prepared by zinc oxide and H2O2?
Studies on the antibacterial characteristics of the oxide of zinc and the output of H2O2 show that the content of H2O2 in the zinc oxide slurry has a direct relation. It means that it is directly proportional to the antibacterial properties of zinc oxide; and the crystal orientation, specific surface area, and composite modification of zinc oxide It has a great influence on the H2O2 production and antibacterial properties in the zinc oxide solution. It is obvious and can be observed that the different forms and properties of zinc oxide have different antibacterial properties and are related to the active oxygen produced.
What is the overall effect of the nano-zinc oxide textile prepared by the original silk modification method on the antibacterial performance?
The original silk improvement method is to mix the antibacterial agent containing zinc oxide nanoparticles with the fiber raw material and then spin the fiber to obtain an antibacterial fiber. The skin-core antibacterial polyester (PET) fiber-containing zinc oxide nanoparticles antibacterial agent is prepared by antibacterial masterbatch, and the sterilization rate of E. coli and the Staphylococcus aureus (S. aureus), that are some species of bacteria exceeds 99%, and zinc oxide nanoparticles uniformly distributed in the fiber interior and the surface, not falling, so the antibacterial PET fibers having had a long sterilization effect.
What is the difference in antibacterial efficacy of textiles prepared with different contents of zinc oxide nanoparticles?
Using sea silk fiber’s ability to absorb metal or metal ions, the antibacterial effect on two kinds of bacteria was studied under the condition of different amounts of zinc oxide nanoparticles. The test results showed that finishing fabrics under the best process prescription inhibited S. aureus. The bacterial rate is as high as 96.43%, and the antibacterial rate against E. coli is as high as 97.58%. It can be made into medical functional dressings and underwear. The anti-inflammatory and wound healing effects have high practical applications in clinical medicine. value.
How does the finishing method prepare zinc oxide nanoparticles antibacterial fabrics, and what are its advantages and disadvantages?
The finishing method generally includes two parts. Since the zinc oxide nanoparticles is easy to agglomerate in the aqueous solution, firstly, a stable zinc oxide nanoparticles dispersion or zinc oxide nanoparticles gel should be prepared, and then a reasonable method should be selected to finish the fabric. zinc oxide nanoparticles antibacterial agent can not only finish pure cotton fabric but also has a good antibacterial effect on other natural fibers. The sol-gel method has the advantages of mild reaction conditions and controllable product morphology. It is indeed become as one of the main and widely used processes for preparing nanoparticles, but it also has some drawbacks: First, the raw materials are expensive, and some organic substances are harmful to humans. Therefore, as a result it makes it priority to find new substitutes; two whole sol-gel preparation process takes a long time, is not suitable for continuous production and processing plant.
What are the applications of zinc oxide nanoparticles in oral treatment?
Some of the applications of preparing colloidal mouthwash from zinc oxide nanoparticles; inducing oral squamous cell apoptosis; compounding with other substrates as oral filling materials; using zinc oxide nanoparticles to construct a new type of antibacterial nano-artificial bone.
As a manufacturer of zinc oxide nanoparticles, what are the development directions?
Explore new comprehensive preparation methods to solve the process defects in existing preparation methods. Further, clarify the doping modification mechanism, break the limitations of the doping application mechanism, and prepare zinc oxide nanoparticles materials that meet various performance requirements. Expand the research field of surface modification and develop ” customized ” surface modifiers to improve the overall physical as well as the chemical characteristics of the zinc oxide nanoparticles.
The characteristics of several preparation methods of nanometer zinc oxide.
Co-precipitation method: The zinc salt and the salt solution doped with ions are slowly added to the precipitant solution, and the uniform dispersion is achieved by stirring. Will sink
The sediment is obtained after filtering, drying, calcining and grinding; the operation is simple and the cost is low.
Uniform precipitation method: After the metal ions are uniformly mixed, the precipitation agent is slowly precipitated in the entire solution through a chemical reaction, and the metal is separatedThe seeds settle down and are obtained after filtering, washing, drying and calcining; the particles are uniform.
Sol-gel method: the organic metal alkoxide or inorganic salt is hydrolyzed to directly form a sol or decoagulated to form a sol, and then the solute is polymerized and gelled, dried, and calcined to obtain the required inorganic material; the particle size is small, and the uniformity is better Good, selective doping, high purity.
Microwave doping method: electromagnetic waves with a frequency of 300KHz-300MHz radiate the object; heating speed is fast, and the general conditions of reaction takin place are mild, High efficiency, small specific surface area along with the comparative small particle size, uniform particle size distribution, and high reaction yield.
It can not only be used as a siIs the zinc oxide nanoparticles hydrogel in the tablet an ideal choice to promote skin healing?
ngle-use adhesive but also can be bonded on different substrates multiple times without significant loss of adhesive strength. Even though the hydrogel has strong adhesion, it is easy to uncover and will not harm the skin, which is very meaningful for meeting actual clinical needs. In addition to excellent adhesion, the hydrogel also has high stretchability, water absorption, antibacterial and biological safety. In vitro cell culture data indicate that the hydrogel is less toxic to cells and will not cause cell death. Rabbit skin defect infection experiments show that the hydrogel can reduce the formation of paralyzed scars. Explain that it has the effect of remodeling the original.

What are the problems in synthesizing sheet-shaped zinc oxide nanoparticles by the hydrothermal method?
The size uniformity of flaky zinc oxide nanoparticles synthesized by the hydrothermal method is poor, and the size cannot be controlled at a level. It is rather way more difficult in order to control the size; acrylamide is a toxic substance, although the toxicity is reduced after polymerization, and then through PB s buffer cleaning can reduce certain toxicity, it is still found to be toxic; it is imperative to establish a set of standard toxicity testing standards suitable for hydrogels. Although zinc oxide nanoparticles hydrogel can reduce the proliferation of paralyzed scars, the specific mechanism of how it affects wound tissues and cells remains to be further studied.
Why is it that the preparation process of zinc oxide nanoparticles particles by the gas-phase method not favored by zinc oxide nanoparticles manufacturers?
The sample prepared by the gas phase method has good regularity and relatively uniform particle distribution, but during the preparation process, the precursor needs to be vaporized or carrier gas is used as the reaction medium, and the reaction parameters need to be strictly controlled, which makes the reaction process more complicated, the output is low, the energy consumption is high, making it very difficult to expand the production of the chemical industry.