Manufacturer Yosoar Is Engaged In The Mass Production Of Copper Oxide
As one of the largest copper oxide nanopowder manufacturers in China, Yosoar has been recognized by many large enterprises in China, many of which are the leaders in different industries.
The volume of CUO is very high every month and can reach 1000 tons/month
Yosoar’s CUO has the advantage of the fast-dissolving speed and low impurity content.
It has the characteristics of high purity, small particle size, uniform distribution, and large specific surface area. It can be used as a colorant for glass and porcelain.
The dissolution rate of copper oxide is very fast, and there is no insoluble matter after dissolution. The chloride ion content must be strictly controlled.
Copper oxide is compounded with other compounds to provide fertilizers, pesticides or herbicides. Later, copper oxide was mainly used as copper fertilizer in total nutrient broth.
It can be used for plastic masterbatch and plastic products. Add 0.2-1% to various substrates.
Nano copper oxide is used as a coloring agent for glass and porcelain, and a polishing agent for optical glass.
Copper oxide is a metal oxide, which can be used as a catalyst for hydrogen oxide or hydrogen peroxide.
Used in PCB electroplating industry
It is a high-grade electroplating material, dedicated to high-end circuit board insoluble anodic acid copper plating process, widely used in mobile phones, notebook computers, storage devices and other electronic and communication products.
When used for PCB electroplating, the first choice is copper oxide, because compared with copper carbonate, it has less impurities, fewer bubbles, and high utilization (copper oxide contains more than 80% copper, and copper carbonate usually has crystals. Water, the general content is much lower than 51.6%).


Used in the antibacterial industry
The main content of copper oxide is copper, because copper is second only to iron and zinc in the human body. It acts as a catalyst when our cells are not oxidized and has an inhibitory effect on viruses and bacteria.
The nano copper oxide produced by our company has antibacterial and antiviral properties. It can be used on medical fabrics, bedding, clothing and socks. It can better prevent bacterial infections and is also the first choice for medical products.
Used in the catalyst industry
As a high-purity nano-copper oxide, it has better catalytic activity and selectivity than ordinary copper oxide powder, and has advantages in light absorption, magnetism, thermal resistance, chemical activity, catalyst and melting point. , Can be used as catalyst and catalyst carrier and battery active material.

Yosoar: Professional Manufacturer Of CUO In China
If your application field is PCB electroplating materials, catalysts, optical glass polishing agents, or ceramic glass coloring, etc., Kunshan Yosoar cuo nanoparticles will be your best choice.
As one of the more advanced this suppliers in China, the powder(CUO) produced by Yosoar is used as PCB electroplating material, catalyst, and copper source for preparing other copper products. It has micron and nanometer particle sizes to meet your product needs.
You can choose our standard particle size copper oxide, or we can customize this with different particle sizes according to customer requirements.
Yosoar includes a skilled oxide technical team with a few years of expertise, that has been trusty by many customers in China. we are able to additionally offer customers with skilled product data answers and application solutions matched.
Don’t hesitate to send us your parameter requirements by email immediately
- Manufacturing
- Certification
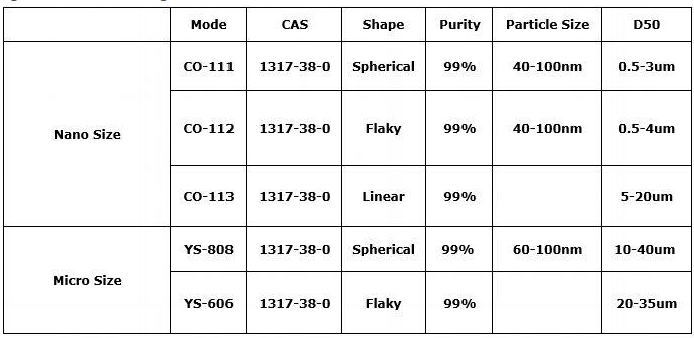
We also have other specifications of copper oxide for you to choose
Copper oxide(CuO) is an inorganic substance. It is a black oxide produced by copper. It is powdery and hygroscopic. Its chemical formula is CuO. It is insoluble in water but relatively soluble in acid.

Regarding how copper powder is produced to generate, Kunshan Yosoar, as the product manufacturer, tells you that the method is actually very simple. You only need to heat the detected copper powder at a high temperature, and a layer of it will be formed on the surface of the copper powder.
Copper oxide powder is a product of copper oxidation. It is generally black, in the form of a black powder, and is an oxide.
Generally speaking, the powder is almost insoluble in water. Generally speaking, insoluble matter mainly refers to that the solubility of this substance in a certain solvent at a certain temperature is very small, because the amount of dissolved is very small, so our The saying is insoluble, but absolutely insoluble matter does not exist.
Regarding the physical properties of the powder, there are several points. First, this is a black powder, and the shape is spherical, flake, dendritic, etc. It is a product that is insoluble in water and alcohol, but it is dissolved in dilute acid, ammonium chloride, etc., and it can also be slowly dissolved in ammonia to form a complex.

The chemical properties of the powder have the following aspects. First, carbon reduces. This reacts with carbon. At high temperatures, it can be reduced to copper and carbon dioxide. Second, this and carbon monoxide are heated under heating conditions. , Can generate copper and carbon dioxide, third, this and hydrogen react, mall copper and water. The last one is the reaction of CUO with dilute sulfuric acid or hydrochloric acid to produce salt and water.
When storing copper oxide nanopowder, keep it in a cool and ventilated place, and keep it away from fire and heat sources. Because it will react with some chemicals, it should be stored separately from reducing agents, alkali metals, and edible chemicals. Don’t confuse it.
When transporting CuO powder, you must first ensure that the packaging of the product is complete. Use special packaging bags for packaging copper oxide nanoparticles chemicals. Ensure that the packaging will not be damaged or leaked during transportation. In addition, oxidation must be prohibited during transportation. For mixed shipments of reducing agents, alkali metals, and edible chemicals that react with copper, protection during transportation must also be protected to avoid exposure to the sun and rain.

As a promising negative electrode material, copper oxide powder has the advantages of easy synthesis, high theoretical specific capacity, high safety, non-toxicity, abundant resources, low cost, and good environmental compatibility. This is the current focus of the development of anode materials for lithium batteries. . The chemical reaction formula of copper lithium storage is as follows: CuO + 2 Li ++2e-⇄Cu+ Li2O
At present, the research direction of this used in anode materials is focused on preparing three-dimensional nanostructured copper oxide particles under alkaline conditions to increase the contact area with the electrolyte, increase the contact surface of the reaction, and improve the reversibility of charging. And discharge. Higher capacitance. Nanostructured electrodes can also make Li + diffusion easier, faster reaction kinetics, more stable structure, and adapt to large strains without severe crushing.
Graphene/CuO composite preparation method At present, based on the preparation method of three-dimensional nanostructured material, the preparation method of graphene/CuO composite material has been synthesized. In the prepared composite material, the copper oxide nanoparticles on the surface of the graphene can be effectively kept adjacent. The dispersibility of graphene sheets and graphene can prevent lithium oxide from agglomerating and crushing during the chemical reaction process of lithium storage. The main preparation methods of graphene/CuO composites are: solvothermal method, one-pot synthesis method, microwave radiation method, self-assembly technology, template method and sol-gel method. 1. Solvothermal synthesis method Solvothermal synthesis method is a chemical reaction that accelerates ion reaction and promotes decomposition under high temperature and high pressure. It is one of the common methods for preparing graphene/CuO composite materials. Researchers add urea (CH4N2O) as a blowing agent during the hydrolysis of copper nitrate to obtain rod-shaped , then add graphene oxide, mix and stir, and then place it in a high temperature and pressure cooker for hydrothermal synthesis reaction to prepare graphene composite material. The porous structure of rod-shaped copper oxide is uniformly attached to the surface of graphene, and CuO nanorods have creeping pores, which accelerate the electron transfer speed of the active material during charging and discharging.
The advantages of the solvothermal synthesis method are: the process is simple, the reaction time is short, and the prepared composite material matrix has good dispersion. The disadvantage is that it is easy to adsorb solution ions. In addition, nano-copper oxides usually grow hydrothermally under alkaline conditions, and the OH concentration is too high, and even Cu(OH)2 precipitates. At present, the research direction of preparing graphene/CuO composites by solvothermal synthesis method tends to add some surfactants to the reaction solution to reduce the appearance of copper hydroxide flocs, and at the same time, it can also adjust the growth rate of different crystals. Plane to obtain a specific structure. Graphene/CuO nanomaterials.

It can be used in photovoltaic cells. Current solar cells are widely used, and it can be used in photovoltaic cells.
Copper oxide is an important p-type semiconductor material with a narrow band gap (1.2-1.5eV) because of its unique characteristics, such as good thermal and photochemical stability, high temperature superconductivity and high electrochemical activity , Non-toxic, cheap preparation method, has been widely used in catalysts, superconducting materials, thermoelectric materials, sensing materials, glass, ceramics, lithium ion batteries and other fields. Currently, because nanomaterials with different morphologies have better optical, electrical, magnetic and thermal properties, the controllable preparation of nanomaterials has become the goal of current materials researchers.

For example, as the most common transition metal oxide, this is gradually applied to lithium-ion batteries due to its high theoretical specific capacity (674mAh·g-1), simple preparation and low cost. In order to improve CuO as a battery, the conductivity of the electrode as a negative electrode material limits the volume change of the carbon-based active material during the charging and discharging of the battery, thereby improving the reversible capacity, cycle life, and charging and discharging stability of the battery. The carbon core shell composite electrode used in lithium ion batteries, the core is carbon fiber, and the shell is a thin layer of copper oxide. The product thin layer has an array-type nano-needle structure and a nano-hole structure.
The nano-needle structure is on the outer surface of the product thin layer, and the nano-hole structure is a hole that penetrates the product thin layer. The preparation method of the carbon core shell composite electrode for lithium ion battery includes: preparation of copper-plated carbon fiber, sintering molding of copper-plated carbon fiber, and surface oxidation treatment of molded copper-plated carbon fiber felt. In the modified composite electrode, the nanoporous structure of the product thin layer facilitates the easy passage of lithium ions in the electrolyte, and then the lithium insertion and delithiation process occurs in the carbon core, thereby increasing the charge and discharge of the carbon nanotubes capacity. Lithium-ion battery; this part is in close contact with the product shell, which not only improves the conductivity of the electrode, but also buffers the volume change during the CuO conversion process; the copper oxide shell tightly wraps the carbon core part, and the nanoneedle structure is very structured It greatly shortens the diffusion distance of lithium ions, increases the effective contact area with lithium ions, and limits the volume of fibers and the removal of lithium during the charging and discharging process of the carbon expansion lithium ion battery, thereby helping to increase the lithium ion The reversible capacity and cycle life of the battery.

This supplier Kunshan Yosoar tells you that the powder is not a dangerous product under normal circumstances, because this is not a flammable product, and it is an odorless powder product, but if eaten, it will cause harm and is a dangerous product.
(CuO) can cause serious damage to the human body. If it is inhaled, It may cause discomfort in the respiratory tract, causing coughing or wheezing. If swallowed, it may cause poisoning because copper oxide reacts with the acid in the stomach to form copper. Copper is a heavy metal among metals and is poisonous. It may cause kidney failure and death in severe cases.
The powder can be used for PCB electroplating, ceramics, and catalysts. It is also widely used in the field of superconductivity. It is also a raw material for making copper compounds.
The supplier Kunshan Yosoar told the stability of this is generally stable. But avoid high temperature and acidic environment.
The product supplier Kunshan Yosoar tells you how copper nitrate turns into CUO. First, an alkali is added to the copper nitrate, and then it reacts to produce copper hydroxide. After heating, the heated copper hydroxide will decompose into this.

Kunshan Yosoar, as a professional copper oxide manufacturer, has a professional technical team to research and produce. The copper content in general is 99%.
(CuO) is not conductive, because it does not contain free electrons and ions that can move, so it is not conductive.
Regarding how basic copper carbonate generates powder, the basic copper carbonate is heated, and the heated basic copper carbonate is decomposed into this, water and carbon dioxide.
Yes,nitrosodimethylamine (NDMA) has a high risk of causing cancer. Researchers in the drinking water industry have increased the risks and concerns of this type of water after it was discovered in a drinking water plant in one country and in a drinking water well near a rocket engine test site in another country. Subsequent investigations showed that it was produced in water after chlorine, chloramine disinfection and ozone oxidation. Some countries in Europe and Asia have also successively inspected source water, wastewater from drinking water plants and wastewater from sewage treatment plants. In view of the widespread existence of NDMA in water bodies, it is necessary to find effective technology to control it.
The reduction technology of zero-valent iron has attracted wide attention due to its wide variety of raw materials and easy operation. It is used to remove pollutants in the environment, including various chlorinated organics, azo dyes, nitroaromatic compounds, etc. Iron can reduce and degrade NDMA, but the reaction time is longer, so it is necessary to find a way to increase the reaction rate. (CuO) is present in the corrosion products of copper pipes for domestic water supply and has been used to catalyze the oxidation of propylene and phenol in water. Methane is dechlorinated, so its application in catalytic zero-valent iron reduction NDMA has great potential.
This is a traditional mineral pesticide. It has the functions of killing insects, sterilizing, fertilizer, wood preservation and regulating plant growth in the field of pesticides. As a fungicide and insecticide, copper has been used in the field of pesticides for a long time, but for traditional copper oxide, there are a series of problems such as slow absorption and poor effect. At this stage, research on copper is mainly focused on nano-copper and photodegradation. Pesticides, organic copper, etc.
In recent years, due to the development of nano-materials, nano-copper oxide also has certain applications in the field of pesticides. Nanomaterials are easier to be absorbed by plants due to their small particle size, which is beneficial to improve the efficacy of medicine. In addition, degradable pesticides have also received more and more attention. Low-toxicity, high-efficiency, residue-free and environmentally friendly pesticides have increasingly become the goal pursued by people. Nano CuO has photocatalytic degradation performance. Using this function, the use of inorganic copper oxide in combination with other fungicides/insecticides can effectively reduce drug residues and improve efficacy, which will become a development trend.

Black copper oxide is a CuO powder conversion film formed on the surface of copper alloys due to the chemical reaction between copper atoms and oxidants.
“Chemical treatment” is a film of a compound formed in the reaction of a matrix material with another material. This reaction distinguishes the conversion coating from the conventional coating applied on the surface of the substrate without changing its chemical state.
Examples of conversion coatings include black oxide on steel, anodic oxidation (the electrochemical process of growing an oxide film on the metal surface connected to the anode in an acid electrolyte) and phosphating treatment (from base metals and phosphoric acid solutions). A coating formed by insoluble crystalline metal phosphate through a chemical reaction between them). For decorative coloring or as an adhesion promoter for polymer overlays,

High-purity CuO has many potential applications due to its excellent physical and chemical properties, including the research fields of catalysis and gas sensors. More advanced applications, such as solar cells, light-emitting diodes, photocatalysts, electrochemistry, electrochromic devices, etc., may be the research field of copper oxide nanostructure materials. In addition, nanostructured high-purity CuO can be used in a variety of applications to remove inorganic contaminants, and can be used as a potential component for manufacturing nanodevices. Nanoparticles specifically designed for antibacterial applications in biotechnology and medicine are in great demand, with a market value of $17.5 billion in 2011.
For hundreds of years, copper has been widely used as an effective fungicide. When exposed to high concentrations of copper, both gram-positive and gram-negative bacteria are killed. As a promising material for preparing antibacterial textiles, high-purity nanoparticles have the same biocidal effect reported in the literature, and have very effective results in cotton fabrics. Recent developments have also reported the antibacterial properties of high-purity nanoparticles used in textiles for biomedical applications. The powder impregnated in cotton or polyester fabric has a good bactericidal effect on various microorganisms and viruses. The low risk of adverse effects on the skin is the advantage of using high-purity copper oxide nanoparticles.

“Due to the high surface area to volume ratio of nanostructured materials, it has a high antibacterial effect, and the dependence of its cytotoxicity on particle size has been reported in the literature. It has also been observed that copper oxide nanoparticles have a high level of cytotoxicity. . Higher than other transition metal oxides (such as iron oxide, titanium dioxide and zinc oxide).
Compared with other metal oxides, their good performance and low cost have aroused people’s interest in the development of copper(II) oxide nanostructured materials. It is believed that its structure is more effective and stable than the antioxidant copper (I) at room temperature.
According to reports, there are many synthetic methods for preparing nanostructured CuO particles, each of which has its own advantages and disadvantages, including sol-gel, hydrothermal, thermal decomposition, microwave radiation, alcohol heating, and heating. Sonochemical process. In this case, this study proposes a method to synthesize and characterize copper(II) oxide nanoparticles using two different precursors. The co-precipitation synthesis method was chosen because of its simplicity, low reaction rate and low cost.
Under the two reaction conditions, sodium hydroxide (NaOH) was used to generate copper oxide nanoparticles in an alkaline medium. The precursors are copper sulfate (CuSO 4 .5H 2 O) and copper chloride (CuCl 2 .2H 2 O). The synthesis was carried out at a temperature of 50°C and 75°C, and the metal salt concentration was 0.1M and 0.2M. After preparation, copper(II) nanoparticles prepared with copper sulfate as a precursor showed better results (smaller crystallite size). The co-precipitation method is considered suitable for obtaining nanostructured materials, and its advantages are simple, fast and cheap.

When using copper(Ⅱ)oxide(CUO), remember to take good care of it. It is best not to contact it with the skin.If you accidentally get it on your skin, rinse it with clean water and use and operate it according to the standard of use.
If CuO leaks, the product manufacturer suggests the following measures must be taken. First, isolate the leaked contaminated area and restrict the access of personnel. For emergency treatment personnel, it is best to wear dust masks and protective clothing. If the amount is small, avoid dust, sweep up carefully, put it in a bag, and put it in a safe place. If the quantity is large, it should be covered with a plastic sheet and recycled to a professional chemical waste disposal site.
Copper oxide powder can be burnt, if the temperature is very high, it can be melted and decomposed into cuprous oxide and oxygen.
The powder supplier Kunshan Yosoar tells you that there are three levels of this product, namely nano-level, micro-level, and industrial grade. According to your different needs, we will recommend suitable product to you.
The main component of (CUO) is copper. Even copper powder contains impurities. Because it comes into contact with air, it also contains impurities.
(CUO) can be used as a catalyst, among which it can be used as a catalyst for producing oxygen from hydrogen peroxide.
(CUO), which is a traditional pesticide of mineral origin, is widely used in the field of pesticides. Copper oxide can play the role of insecticide and sterilization, and the copper element in copper oxide can also supplement the copper element required by plants.
Kunshan Yosoar has a dedicated factory, so the copper oxide of Kunshan Yosoar is shipped from Kunshan Yosoar’s factory and sent to customers.
Kunshan Yosoar manufactures. Because the factory provides products at any time, considering that the customer needs to test for the first use, the minimum order quantity is 1kg.