Copper Oxide Nanoparticles High Quality
Manufacturer Yosoar Is Engaged In The Mass Production Of Copper Oxide Nanoparticles
Exposure of CuO NPs to living systems can lead to the production of reactive oxygen species.
Nano copper oxide can be used in sensors, semiconductor manufacturing, and other electrical applications.
Nano-sized copper oxide has different physical properties about 100 times thinner than copper oxide.
It has photovoltaic and photoconductive properties.
Nano-copper oxide is often used in the form of a polymer coating.
Used in farmland soil
Nano-copper oxide can affect the soil by reducing the nitrification, denitrification and soil respiration that occur in the soil, which also has an impact on plants.
During nitrification, soil ammonia is oxidized to nitrate (NO 2-), and then oxidized to nitrite (NO 3 -). The conversion of ammonia to nitrate is carried out by autotrophic bacteria and Achaea, and it is converted to nitrite by Nitrobacter and Nitrobacter. Nitrospira is complete.
These two processes are necessary for the final release of nitrogen, which allows plants to produce proteins that are essential for their growth. Excessive use of nano-copper oxide in the soil can inhibit or reduce the production of important nitrates. If it is in rough and medium-textured soil, it will be more serious. Nano particles are the most toxic to nitrifying bacteria.

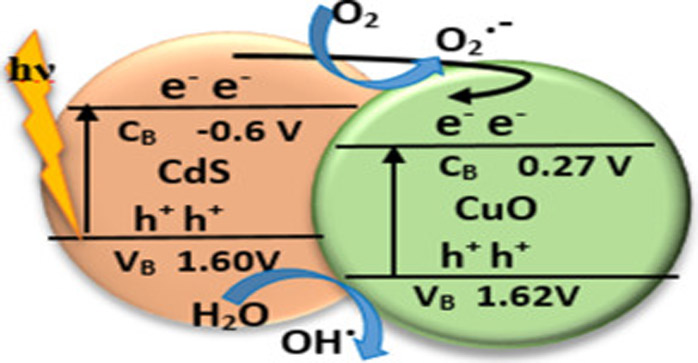
Used in Photocatalytic
Nano-copper oxide has photocatalytic activity. For example, under ultraviolet light, the photocatalytic degradation activity has an effect on crystal violet and methylene blue dye.
CuO NPs (ie 0.03mg CuO NPs) were added to 30mL CV dye solution in a 50ml glass beaker. The CuO NP/dye solution was placed under stirring at a set distance from the UV lamp (365nm wavelength: 0.7AMPS). The applied wavelength range is 190-950 nm. The degradation efficiency of CuO NPs on CV dyes after 36 hours of UV irradiation is 92.126%. CuO NPs effectively degrade organic dyes in aqueous solutions, opening up a wide range of photocatalytic applications.
Yosoar: Professional Manufacturer Of Copper Oxide Nanoparticles In China
- Manufacturing
- Certification
The overall particle size of the CUO nanoparticles ranges between 1-100nm. It exhibits peculiar physical and chemical properties in terms of magnetism, light absorption, chemical activity, thermal resistance, catalyst, and melting point.

copper oxide nanopowder are a black to brownish-black amorphous powder. The melting point of this is about 1330°C. It dissolves in acid to form copper salt, dissolves in ammonia, ammonium chloride, and potassium cyanide solution to form soluble complex salts respectively, and also dissolves in dilute alkali metal hydroxide solution, and then the color will change to blue. Apart from this, it is not soluble in water and ethanol and reacts with acetylene and berkelium.
Brief introduction of CUO nanoparticles
Copper oxide powder in its general appearance is a brown-black metal oxide powder and has many applications and is widely used in various fields. Ordinary copper oxide is a multifunctional fine inorganic material with a wide range of uses, mainly it is used in the printing and dyeing processes, also in manufacturing glass, ceramics, medicine, catalysis, and other fields as well. Apart from this, it is also generally used as a catalyst and as a catalyst carrier as well along with its use in electrode activation material, and also as a rocket propellant. As being one of the main and integral parts or components of the catalyst, this oxide powder has vast applications and is used in a variety of catalysts for oxidation, hydrogenation, NO, CO, reduction, and hydrocarbon combustion. Also, it is very widely utilized in the reaction. The powder also has superior activity in regards to the catalytic and selectivity and other properties when related or compared to the large-size copper oxide powder.
—Particle size
The particle size of copper oxide nanoparticles is between 1-100nm. Compared with ordinary copper oxide, it has special electrical, optical, catalytic, and other properties.
—Electrical properties
The electrical properties of CUO nanoparticles generally enable them to be much more sensitive to external environments including factors such as those related to temperature, humidity, or light, etc. Therefore, the use of CUO nanoparticles particles to coat the sensor can generally result in an apparent increase and an overall improvement to the response speed, general sensitivity, and overall selectivity of the sensor.
—Spectral characteristics:
The spectral properties of CUO nanoparticles show that its infrared absorption peak is obviously broadened, and there is an obvious blue shift phenomenon. The nanometer preparation of copper oxide found that CUO nanoparticles with the smaller size of the overall particles and better dispersibility and also have higher functioning and performance when compared to the ammonium perchlorate.

The general appearance and size and shape of nanoparticles significantly affect the properties and uses of nanopowder and its products. Therefore, nanopowder measurement technology has an important role to be played in the general research and development of nanopowder technology. A variety of particle size measurement methods have been developed, and these methods have been continuously improved and perfected in practice. The analysis and testing methods of common powder and the corresponding physical and chemical characteristics are as follows:
—Transmission electron microscope (TEM): overall appearance and internal structures and size of the particle size
—X-ray diffraction (XRD): the crystal form and size of the particles
—Scanning Electron Microscope (SEM): the morphology and size of aggregates
—Atomic emission spectroscopy (AAS): qualitative and quantitative analysis of particle chemical composition
—Infrared spectroscopy (IR): particle intermediate structure, particle structure
—X-ray fluorescence (XRFS): chemical composition of particles
—X-ray small-angle scattering method (SAXS): particle size
—Thermogravimetric analysis (TG): the reaction mechanism of desorption and decomposition of adsorbed substances on the surface of particles
—Differential thermal analysis (DTA): the crystal form transition temperature of the particles
—EDTA chemical analysis: particle chemical composition, CuO content
—BET method: specific surface area of particles
copper oxide nanoparticles have a variety of crystalline states with different morphologies. The following figure shows three common this crystal morphologies.
Rod crystal nano cuo
Needle crystal copper oxide
Flake crystal copper oxide
—The powder is prepared by high-frequency plasma gas-phase combustion method. The overall characteristic of the product includes high purity, relatively small particle size, and overall uniform distribution, and large specific surface area, an overall high activity on the surface, low bulk density, and the general gas phase method preparation, which overcomes the wet chemical method on the market the prepared particles have shortcomings such as hard agglomeration, difficult to disperse, and low purity.
—They are soluble in dilute acid, NH4Cl, (NH4)2CO3, potassium cyanide solution, insoluble in water, and slowly dissolving in alcohol and ammonia solution. It can also be minimized to metallic copper generally when it meets hydrogen or carbon monoxide at high temperature.
—The powder has better catalytic activity, selectivity, and other application properties than large-size copper oxide powder.

—The usage of copper oxide nanoparticles in catalyst
Cu is the 29th element. It has some quite amazing properties including an overall special electronic structure and gains and losses electrons and exhibits a good catalytic effect. This is because of the overall large surface area, high reaction activity, no pollution, and strong selectivity, they have shown good catalytic effects in many reactions.
The application of ultra-fine nano-scale catalysts is indeed an important solution for adjusting the combustion performance of propellants. It is also used in the sector of national defense, ammonium perchlorate (AP) is the high-energy component of composite solid propellants. It occupies 60% to 80% of the AP propellants. Needle-shaped copper oxide having the size of about 30-50 I prepared by spray pyrolysis is linked with the general surface of AP crystals to form composite particles, thereby reducing the thermal decomposition temperature of AP, accelerating the decomposition rate, and the total heat of decomposition will increase.
—The usage of nanoparticles of copper oxide in antibacterial agent
The powder has a loose porous spherical morphology, and the extremely low-concentration it have good antibacterial effects on Escherichia coli, Pseudomonas aeruginosa, Salmonella, Bacillus subtilis, and Staphylococcus aureus. When the light energy is greater than the bandgap of this(1.2eV), electrons can be excited from the valence band to the conduction band, generating corresponding electron-hole pairs, and reacting with H2O or O2 in the environment to generate strong oxidizing hydroxyl radicals ( • OH) and superoxide anion (O2-) and other active groups. These active groups can penetrate the cell wall of bacteria, chemically react with the organic molecules in the cell, destroy the cell membrane structure, cause the death of the bacteria, and achieve the purpose of antibacterial.
— The application of copper oxide nanoparticles in desulfurization agent
At present, a large amount of sulfur-containing gases, such as hydrogen sulfide and sulfur oxides, are emitted into the atmosphere during industrial production, causing serious air pollution. Tests have verified that some metal oxides have good desulfurization properties, including CUO nanoparticles, and have good desulfurization effects. copper oxide nanoparticles with different grain sizes have different desulfurization effects; overall the appearance of oxygen vacancies that are observed on the surface of the product and the decrease of electron density around the copper element is beneficial to improve the desulfurization performance; when the difference in specific surface area of it is small, the desulfurization activity The effect of sulfide is not significant, but if the irregular pore distribution formed by the accumulation of particles is narrow, and there are both open and contracted pore structures, it is beneficial to the removal of hydrogen sulfide.
Nanoparticles of copper oxide are indeed excellent desulfurization products, this is because they can exhibit excellent activity at normal room temperature, and ultimately the removal accuracy of H2S reaches below 0.05 mg•m-3. After optimization, a change is observed in the overall penetration sulfur capacity of copper oxide nanoparticles and it reaches 25.3% at a space velocity of 3000 h-1, which is a bit higher when compared to other similar desulfurization products.
—The application of copper oxide nanoparticles in sensors
In terms of sensors, these nanoparticles of copper oxide can also be used as the coating film of the sensor to improve the sensitivity and selectivity of the sensor to carbon monoxide gas.
A physical sensor as the name indicates is a device that changes or converts general physical quantities such as light, sound, magnetism, or temperature from the outside into electrical signals. Chemical sensors are devices that convert the types and concentrations of specific chemical substances into electrical signals. It is mainly to design chemical sensors by directly or indirectly causing changes in electrical signals such as electrode potential when sensitive materials and molecules, ions, etc. in the measured substance are in contact with each other. Because the external signal can be converted into an electrical signal in order to indicate, the sensor is also very widely used in many fields related to environments such as environmental monitoring, medical diagnosis, and meteorology.
copper oxide nanoparticles have the advantages of high specific surface area, high surface activity, and specific physical properties. It is very sensitive to the external environment such as temperature, light, humidity, and certain specific volatile gases. It has excellent response speed, sensitivity, and Selective.
—The application of CUO nanoparticles in batteries
In the battery industry, it can be used as the cathode material of solar cells and the anode material of lithium batteries.
—The application of copper oxide nanoparticles in other areas
At present, the application of CUO nanoparticles in other aspects is also very extensive. They are also used in ceramic additives or colorants, exhaust gas purification materials, anticorrosive paints for wood products, hydrogen storage materials, and contact materials.

Copper oxide and cuprous oxide belong to the general mineral-derived pesticides, which have the basic function in terms of killing the insects, sterilizing, fertilizer, wood preserving, and apart from these also regulating the overall plant growth in the field of pesticides. Although we do know that copper has indeed got a long history of being a fungicide and insecticide in the field of pesticides, despite the fact, the traditional inorganic copper oxide has a series of problems. These problems include a relatively slow absorption and obviously a poor effect. At this stage, the research on copper mainly focuses on CUO nanoparticles and organic Copper, and other aspects.
Recently we have observed that there has been development in the relevant field and because of this development of the nano-materials, the importance of copper powder has also been improved. The nanoparticles of copper oxide also have certain applications and usages in the field of agriculture, especially in the pesticides field. Now we know that nanomaterials are easier in being getting absorbed by the plants, and this is only because of the small particle size of these materials, which is generally more conducive to the improvement and effectiveness of drug efficacy. In addition to this, there also has been an increase in the sense of giving attention to degradable pesticides. Having low toxicity, higher rate of efficiency, being residue-free, and environmentally friendly pesticides are becoming an increasing concern and many people have made it an aim in order to achieve this. It also have photocatalytic degradation properties. This feature enables us for compounding inorganic copper oxide with other fungicides/insecticides and can also effectively reduce drug residues and therefore, it results in improving the overall efficacy, which is definitely going to become a development trend.
The nanoparticles of copper ii oxide antibacterial agent are added in various synthetic resins as well and these synthetic resins include PP, PE, PVC, PS, PET, ABS, etc., apart from this, various coatings, some adhesives, various ceramic well as artificial stones, and synthetic fibers. This adding of the nanoparticles of copper oxide antibacterial agent to synthetic resin or fiber and the masterbatch can be varied. This means that you can select it according to the required or the specific production conditions. In a similar way, the nanoparticles of copper oxides antibacterial agent can also be added to the coating, adhesive and artificial stone, etc., and you can also directly add the powder formulation as well.
—If we talk about the use of antibacterial agent in the ceramics and enamel industry, then the product is basically added to the glaze at a ratio of 0.5%, grind, polish, and fire together.
—When using this antibacterial agent in plastics, the product is firstly added into the raw material of plastic at a ratio of 0.2-0.4%, and then it can be injected or molded, extruded, and can even be sprayed together with the plastic raw materials.
—When using this antibacterial agent for coating, the product, and the coated inorganic material can then be even ground and dispersed together at a ratio of 0.2-0.4%.
—When using this antibacterial agent as dry chemical fiber, the product is firstly added to the raw material of the chemical fiber at a ratio of 0.3% that leads to drawing the metal wires together.

—The form of powder antibacterial agent: this product is generally present as black powder
—If we talk about the average particle size of these nanoparticles of copper oxide antibacterial agent, then it is 1~2μm
— The overall specific surface area of this antibacterial agent is around 10-15m2/g
—The general persistence of this antibacterial agent is about 600℃
—Whereas the antibacterial activity of antibacterial agent is regarding the inhibition rate against Escherichia coli, staphylococcus aureus, and candida Albicans is over 99%
—Nanoparticles of copper oxide that act as the antibacterial agent have antibacterial function for plastic products and these products include refrigerators, plastic shell, inner liner, telephone case, keyboards of computers, plastic parts of washing machines, power plastic switch, water pipes made of plastic, cutting boards, seat, fruit Plates, children’s toys, etc. and many more.
—antibacterial agent also has a variety of antibacterial functions that are involved in the daily use of enamel, ceramics, and glass products. These include products such as bowls and plates, some of the sanitary products, utensils used for drinking, etc.
—antibacterial agent has many antibacterial functions, some of these include their use in the fiber, bedding, medical and sanitary products, underwear, etc.
—antibacterial agent has a variety of antibacterial and deodorant functions, such as lining, surface lining, insoles, etc.
—antibacterial agent is also very much suitable for various packaging materials. These packaging materials may include various plastic packaging bags, food packaging boxes, film bags, etc.
—antibacterial agent is also a great option to be used in health-related fields or the overall medical sector. These can be used as disposable syringes, infusion sets, isolation clothing, etc.
—antibacterial agent broad-spectrum antibacterial
antibacterial agent is a broad-spectrum antibacterial agent with long-lasting bactericidal and antibacterial effects. Further, we can say that it is harmless to the general body of humans and it also causes no irritation to the skin. It is indeed a safe and harmless product.
—antibacterial agent with high weather resistance
antibacterial agent in terms of stability is highly chemical stabile and also in terms of thermal stability as well, this can be understood from this that it can even withstand the high temperature of 300℃, and even at such temperatures, it will not change color much easier.
—High stability of the antibacterial agent
After surface treatment, it can be mixed with various resins and even in such conditions, it has no effect on the overall processing performance and other general properties of the material as well.
—antibacterial agent continues to be antibacterial
antibacterial agent can be permanently combined with high-scoring materials, is resistant to acid and alkali, is resistant to washing, is not easy to produce bacterial resistance, releases slowly, and has a long-lasting sterilization effect.
—antibacterial agent has strong adaptability
antibacterial agent can also be powder surface treated. This could be done in accordance with the user requirements so that in order to achieve dispersion in the overall medium of the matrix.
—antibacterial agent is easy to use
an antibacterial agent can also be made into an antibacterial masterbatch or dispersant most generally for fibers, plastics, paints, fabrics, etc. It is convenient to use in this regard.
—The first method is to directly prepare particles and disperse them in the required solvent to obtain a dispersion of copper oxide nanoparticles, and then form a film by spin coating or other methods. This method has the following advantages: (1) You can choose a suitable solution for dispersion; (2) Since it have been formed, high-temperature annealing or even annealing is not required, and it is suitable for flexible devices. However, this method also has certain drawbacks. For example, the poor dispersion of copper oxide in the solution leads to many defects in film formation and lower device efficiency.
—The second method is to prepare the precursor solution of copper oxide nanoparticles, spin-coated the precursor solution into a film, and fire it into copper oxide nanoparticles by calcination. If we talk about the overall advantage of this method, then it is that the dispersion of the precursor liquid is good, and the efficiency of the prepared device is high. However, this method requires high-temperature calcination, the required cost is relatively high, and its scope of application is limited.

—Preparation of nanometer copper oxide by the precipitation method
—Preparation of nanometer copper oxide by the complex precipitation method
—Preparation of nanometer copper oxide by the reflux precipitation method
—Preparation of CUO nanoparticles by the hydrothermal method
—Preparation of CUO by the sol-gel method
—Preparation of nanometer copper oxide by the microemulsion method
—Preparation of CUO by the laser evaporation method
—Preparation of nanometer copper oxide by the template method
CUO nanoparticles are prepared by the precipitation method. If we consider this general method, then it includes the use of a metal copper salt and a precipitating agent undergoes in a reaction in a solution according to a certain ratio to produce a precipitate, and the prodcut are obtained through aging, washing, drying, and calcination. The precipitation method has the advantages of simple method, low cost, narrow size distribution, high yield, easy promotion, and industrial production.
copper oxide nanoparticles are prepared by the complex precipitation method. In the complex precipitation method, a copper salt is firstly reacted with a complexing agent to form a complex, and then reacted with a precipitating agent to obtain a precursor, and then heat-treated to obtain copper oxide nanoparticles. The main synthesis method is to first prepare Cu(NO3)2 into a solution. Under a certain temperature and full stirring, slowly add the complexing agent dropwise to gradually generate the copper complex, and then dropwise add the precipitating agent under constant stirring. So after the completion of the reaction, filter and wash the precipitate. The precipitate is first washed with distilled water and then with ethanol, and then vacuum-dried at 80°C for 2 hours and heat-treated at 400°C for 2 hours to obtain this.
What is the reflux precipitation method to prepare cuo nanoparticles?
copper oxide nanoparticles are prepared by the reflux precipitation method. The reflux precipitation method basically refers to the formation of manufacturing of nanoparticles by ordinary or microwave heating and boiling reflux. The main steps are: take a certain amount of copper nitrate, dissolve it in water, add alkali to adjust to a predetermined pH value, stir and heat to boil and reflux for 1 hour to obtain a dark brown precipitate, filter it while hot, wash it with distilled water, and dry it in an oven at 80°C for 5 hours Above, the dark brown powder can be obtained.

What is the hydrothermal method to prepare copper oxide nanoparticles?
copper oxide nanoparticles are prepared by the use of the hydrothermal method. The hydrothermal process generally uses water as the reaction solvent. It is a method of nucleating, growing, and recrystallizing copper oxide nanometers by nucleating, growing, and recrystallizing metal copper salt and alkali solution in a special closed reactor at a relevant high temperature as well as a high pressure. For example, using Cu(CH3COO)2•H2O and NaOH as raw materials, a simple hydrothermal method is adopted for a one-step reaction to prepare the particles with high yield and uniform size.
The hydrothermal method, also known as a hydrothermal method, belongs to the category of liquid phase chemistry. It means that the solubility of some hydroxides in water under high temperature and pressure is greater than the solubility of corresponding oxides in water, so the hydroxides are dissolved in water and oxidized at the same time Method of precipitation. The hydrothermal method is characterized by low production cost, high particle purity, good dispersion, good crystal form, and controllability.
What is the sol-gel method to prepare copper oxide nanoparticles?
cuo nanoparticles are prepared by the sol-gel method. The sol-gel method is also known as colloidal chemistry. It is to hydrolyze and condense the reactants to form a stable transparent sol system, and then concentrate to form a transparent gel with a three-dimensional network structure. After drying and heat treatment, copper oxide nanoparticles particles can be obtained. The method has a uniform distribution, small particle size, high purity, and can be industrially produced, but the raw material cost is high, also the washing is difficult, and apart from this, the agglomeration during drying is way easier.

The basic steps of preparing copper oxide nanoparticles powder by using the sol-gel method combined with supercritical drying technology are: dissolve the prepared copper salt solution (Cu(NO3)2) in absolute ethanol, and put the ethanol solution of copper salt in ethanol. In a human autoclave, program the temperature rise and control the pressure (the temperature and pressure should be higher than the critical temperature of ethanol 243℃ and the critical pressure 6.38MPa respectively), keep the pressure for a period of time; then slowly deflate, and then ventilate the protective gas (N2) Naturally cooled to room temperature, black fluffy powder is prepared. Using this method, particles with a uniform particle size distribution, no obvious agglomeration, along with an average size of the particle of about 10 nm can be obtained.
What is the preparation of copper oxide nanoparticles by the microemulsion method?
cuo nanoparticles are prepared by the microemulsion method. The microemulsion method is a method in which two immiscible solvents are used to form an emulsion under the action of a surfactant, and nano-particles are obtained after nucleation, coalescence, agglomeration, and heat treatment in microbubbles. Microemulsions are usually transparent and isotropic thermodynamically stable systems composed of surfactants, co-surfactants (usually alcohols), oils (usually hydrocarbons), and water (or aqueous electrolyte solutions). The product prepared by the microemulsion method are small in size and controllable, and the reaction conditions are easy to achieve, but the amount of surfactants and solvents consumed is large, the cost is relatively high, and the overall selection of the system is a little bit difficult, and the operating conditions are demanding.
What is the preparation of copper oxide nanoparticles by laser evaporation?
copper oxide nanopowder are prepared by the laser evaporation method. The laser evaporation method uses a laser as a heat source to decompose and evaporate the copper salt and then condense it to form nanoparticles. The steps are as follows: the reactant absorbs the energy of the laser and quickly vaporizes to break the chemical bond, and then reacts to form nanoparticles. The preparation process is to directly irradiate the focused laser beam to the cylindrical solid copper acetate target in the reactor. The copper acetate absorbs the laser to heat up, decomposes, and evaporates, and the steam quickly cools down and condenses into nanoparticles, which enter the trap with the carrier gas. It is collected in the vessel. The laser evaporation method has a short reaction time, fast speed, good dispersion of nanoparticles, small particle size, uniform distribution, and almost a single spherical shape, controllable reaction, simple and continuous process, large output, and production The cost is low, and it has good industrial application prospects.
What is the template method to prepare copper oxide nanoparticles?

CUO nanoparticles are prepared by the template method. The template method uses molecular sieves, membranes, or nanotubes as templates in the reaction to prepare one-dimensional nanostructured nanoparticles.
Using carbon nanotubes as a template, rod-shaped cuo nanoparticles particles were prepared, this was done with a particle size ranging from 20 nm to 90 nm along with the length of 250 nm to 700 nm. The main process is to prepare polyhedral carbon nanotubes, then weigh a certain amount of the carbon nanotubes, heat them with a nitric acid solution for 24 hours, wash with water, and dry at the temperature of 60°C for 24 hours. Add the treated carbon nanotubes to 50 mL saturated nitric acid. In the copper solution, after reacting for 48 h under stirring, filter, and wash, the sample was dried at 60℃ for 12 h, then calcined in an argon atmosphere for 5 h, and finally calcined in air at 750℃ for 1 h. The carbon nanotubes are removed to prepare copper oxide nanoparticles.
This preparation method firstly needs to prepare carbon nanotubes, which is costly. However, the particle morphology core size can be controlled, the product size distribution range is wide, and the yield is high. This is a good method for preparing one-dimensional particles.
Why do we usually need to introduce ultrasound when preparing copper oxide nanoparticles?
Ordinary copper oxide present is indeed an important industrial product, which has a wide range of applications in the fields of glass, ceramics, printing, and dyeing, and medicine. When it reaches the nanometer level, it has a quantum size effect, surface effect, etc. And is indeed one of the most widely used inorganic materials, the preparation method of copper oxide nanoparticles is also very important.
We all are aware that ultrasound is a sound wave with a frequency higher than 20000Hz. It has good directivity, strong penetrating ability, easy to obtain concentrated sound energy, and spreads far in the water. The special acoustic cavitation effect of ultrasound provides an important way to prepare materials with special properties. The source of the acoustic cavitation effect in ultrasound is the growth, contraction, and collapse of the tiny bubble nuclei in the system, which leads to local instantaneous high temperature and high pressure, accompanied by strong high-speed micro jets, shock waves, and discharge luminescence. These effects can be controlled. The overall size and the distribution of the particles as well promote the formation of new solid phases and prevent the aggregation of nanoparticles. And because of its advantages such as simple operation, easy control, and high efficiency, ultrasonic technology is widely used to assist the preparation of nanomaterials and has achieved good results.
In the preparation of CUO nanoparticles, the ultrasonic-assisted method has become an effective method. The assistance of ultrasound can refine particles, promote particle crystal transformation, and improve certain properties of copper oxide nanoparticles.
The preparation of CUO nanoparticles by the ultrasonic-assisted method breaks through the traditional preparation method. The ultrasonic technology is not only easy to operate but also effectively solves the problem of agglomeration of nanoparticles and promotes the refinement of crystal grains. Similarly, an important transition metal oxide semiconductor material, the excellent performance of CUO nanoparticles also make it show a very broad application prospect in many fields.

What are the prospects for copper oxide nanoparticles?
We see that copper oxide have indeed got a wide range of applications whether it is used as a catalyst in any reaction or as a different type of material because the preparation method is simple, the raw materials are easily available and other factors have attracted many people’s attention. We can also see the development direction of modern chemistry from the preparation of the nanoparticles of copper oxide. Although the raw material formulations are different and the process parameters are different, we are all using the latest scientific and technological methods in order for developing some new methods and new processes to achieve energy saving, high efficiency, and green Production, and then realize the industrialization of production.
In view of the huge application potential of copper oxide nanomaterials in many fields, the preparation and application research of nanoparticles of copper oxide will still be very active in the future. As a new type of material, it has a very wide range of applications, because the preparation method is simple, the raw materials are easily available and other factors have attracted many people’s attention. At present, the most important problem to be solved for the industrialization of the production of it is the problem of particle agglomeration. The prospects for copper oxide are as follows:
—Research on the anti-agglomeration technology of copper oxide nanopowder
It is easy to agglomerate due to their small particles, large specific surface area, and high surface activity. If the agglomeration problem cannot be solved, it will be difficult in order of making full use of the characteristics of this.
—Research on the microstructure control technology of copper oxide nanoparticles
Exploring different chemical and physical preparation methods and revealing the laws that control the microstructure, size, and growth morphology of copper oxide nanopowder will be an important research field of copper oxide nanomaterials.
— Research on the relationship between the microstructure on the nanoparticles of copper oxide and its properties
Because the function of nanomaterials not only depends on the chemical composition of nanomaterials but also on the morphology and physical composition of nanoparticles. Therefore, research on the relationship between the microstructure of this and their properties is also important and is the key technologies that need to be resolved.
What technical support do we provide?
We provide technical support for the application of copper oxide nanoparticles in catalysis and additives. For specific application inquiries, please contact the sales staff.

How to package and store copper oxide nanopowder?
The packaging of this is inert gas packaging. Therefore, you should seal it in a dry environment and the place should also be cool. Further, these should not be given exposure to the air for a relatively long time so that the agglomeration of this could be prevented that is caused because of moisture, as this generally affects the overall dispersion performance and use effect.
The human body safety precautions of copper oxide nanoparticles
copper oxide nanopowder are harmful to the human body.